Press Release (ePRNews.com) - DELHI, India - Apr 11, 2017 (UTC) - The report entitled “Global Long Acting Injectable Anti-psychotic Market: Size, Trends & Forecasts (2017-2021)”, provides analysis of the global injectable anti-psychotic market, with detailed analysis of market size and growth, and segmentation of the industry.
Under competitive landscape, different products in the long acting injectable anti-psychotic market have been compared on the basis of different parameters such as dosing frequency, patent expiry, injection sites, label, storage, needles and available strengths. Also, a financial comparison of long acting injectable market players in the US has been done.
Moreover, the report also assesses the key opportunities in the market and outlines the factors that are and will be driving the growth of the industry. Growth of the overall global long acting injectable anti-psychotic market has also been forecasted for the years 2017-2021, taking into consideration the previous growth patterns, the growth drivers and the current and future trends.
Johnson & Johnson, Eli Lily and Company, Alkermes Public Limited Company and Lundbeck are some of the key players operating in the global long acting injectable anti-psychotic market, whose company profiling has been done in the report. In this segment of the report, business overview, financial overview and business strategies of the companies are provided.
Country Coverage
The US
Company Coverage
Johnson & Johnson
Eli Lily and Company
Alkeremes Public Limited Company
Lundbeck
Executive Summary
Anti-psychotic refers to a class of medicine used for the treatment of psychosis and various other emotional and mental conditions. Psychosis consist of delusions, hallucinations, paranoia and disordered thought. It is a condition in which a person experiences thought disorder and personality changes. Long acting injectable anti-psychotics are a sub type of antipsychotics which were primarily developed to improve the treatment of schizophrenia.
The global long acting injectable anti-psychotic market can be studied on the basis of different products and on the basis of prescription. These products are different types of long acting injectable anti-psychotic namely Invega Sustenna/ Trinza, Risperdal Consta, Abilify Maintena, Zyprexa, Aristada and many more.
The global long acting injectable anti-psychotic market is expected to increase at a healthy rate during the forecast period (2017-2021). The global long acting injectable anti-psychotic market is supported by various growth drivers, such as increasing cases of schizophrenia, rising number of bipolar disorder patients and non adherence to oral anti-psychotic.
However, side effect associated with anti-psychotic and patent expiry of some of the leading brands are some of the challenges faced by the market. Aging dernography, grant of approval to Trevicta in European Commission, third party proprietary technologies and label expansion of Abilify Maintena are some of the latest trends in the global long acting injectable anti-psychotic market.
List of Figures:
Figure 1: Psychiatric Disorder
Figure 2: Global Anti-psychotic Market by Value; 2011-2016 (US$ Billion)
Figure 3: Global Anti-psychotic Market by Value; 2017-2021 (US$ Billion)
Figure 4: Global Anti-psychotic Market by Products; 2016
Figure 5: Global Anti-psychotic Market by Prescription; 2016
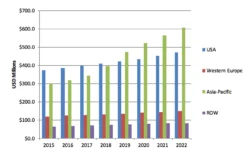
Figure 6: Global Long Acting Injectable Anti-psychotic Market by Value; 2015-2021 (US$ Billion)
Figure 7: Global Long Acting Injectable Anti-psychotic Market Value by Product; 2016
Figure 8: Global Long Acting Injectable Anti-psychotic Market Volume by Products; 2016
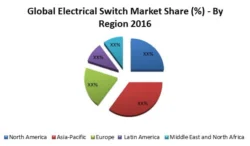
Figure 9: Global Long Acting Injectable Anti-psychotic Market by Region; 2016
Figure 10: Global Abilify Maintena Market by Value; 2015-2021 ( US$ Million)
Figure 11: Global Aristada Market by Value; 2015-2021 (US$ Million)
Figure 12: Global Invega Sustenna Market by Value; 2015-2021 (US$ Billion)
Figure 13: Global Risperdal Consta Market by Value; 2015-2021(US$ Million)
Figure 14: Global Zyprexa Market by Value; 2016-2021(US$ Million)
Figure 15: Global Zyprexa Market by Region; 2016
Figure 16: The US Long Acting Injectable Anti-psychotic Market by Value 2016-2021 (US$ Billion)
Figure 17: Schizophrenia Patients in the US;2015-2025 (‘000)
Figure 18: Bipolar Disorder Patients in the US; 2017-2025 (‘000)
Figure 19: Global Population (65 years and Above); 2011-2015 (Million)
Figure 20: US Long Acting Injectable Antipsychotic Market by Players; 2016
Figure 21: Johnson & Johnson Sales; 2012-2016 (US$ Billion)
Figure 22: Johnson & Johnson Sales by Segments; 2016
Figure 23: Eli Lily and Company Revenues; 2012-2016 (US$ Billion)
Figure 24: Eli Lily and Company Revenues by Segments; 2016
Figure 25: Alkermes Revenues; 2012-2016 (US$ Million)
Figure 26: Alkermes Revenues by Region; 2016
Figure 27: Lundbeck Revenues; 2012-2016 (US$ Billion)
Figure 28: Lundbeck Revenues by Products; 2016
Table 1: Side Effects of Injectable Anti-psychotic
Table 2: Antipsychotic Patent Expiry Dates
Table 3: Global Long Acting Injectable Antipsychotic Market Products Compariso
For further details, kindly visit :
http://www.daedal-research.com/global-long-acting-injecta…
Rajeev Kumar
(Business Development Manager)
Address: 36 SFS Flats
Paschim Vihar
New Delhi-110063
Mobile: +91-9811715635
Tel: +91-120-4553017
Mail ID – info@daedal-research.com
Source : Azoth Analytics