Press Release (ePRNews.com) - PITTSBURGH - Jun 15, 2017 (UTC) - ,
Introduction
TCEs allow light to be transmitted through solar cells and touch screens by forming the electrode necessary for operation. TCEs stand for transparent conductive electrodes. Currently, TCEs use indium tin oxide having low resistance and high transmittance, but if bent lose electrical characteristics. Alternative materials include silver NWs sprayed on polymers offering low-cost fabrication while maintaining electrical properties when subjected to mechanical deformation. NWs stand for nanowires.
However, silver NWs on thin polymer substrates like PET are found to yellow. PET stands for polyethylene terephthalate. The yellowed image noticeably differs from the actual color and since the color of the image is important, efforts have been made to eliminate yellowing. One approach is to coat the silver NWs with hydrazine [1] on the premise any ambient light absorbed by the NWs is not reflected. For silver NWs, SPRs occur in the violet (350 to 390 nm) and thought to absorb ambient light up to 500 nm. Hence, yellow light at (570 to 590 nm) would be observed as reflected light. SPR stands for surface plasmon resonance.
Problem
Silver NWs in TCEs on a PET substrate are observed to yellow, but do not appear orange (590 to 620 nm) or red (620 to 750 nm) even though all ambient light reflected beyond the yellow (λ > 590 nm) should also be observed. Moreover, TCE yellowing under normal operation lacks a mechanism to excite the SPRs at VIS frequencies in the violet and molecular states of hydrazine in the IR suggesting SPRs without a source and absorption of ambient light cannot be the explanation for TCE yellowing.
Proposal
Yellowing of TCEs is caused by QED creating EM radiation at EUV levels in NWs from Joule heat. EUV stands for extreme ultraviolet. Since EM energy in the EUV is greater than that for SPRs in the VIS and molecules in the IR, the SPRs and molecules are excited by fluorescence as the EUV radiation is lowered to VIS and IR levels by radiationless processes. QED stands for quantum electrodynamics, but is a simple form of the complex light-matter interaction proposed by Feynman and others.
Theory
QED radiation is a consequence of QM that precludes atoms in NWs from having the heat capacity to conserve Joule heat by an increase in temperature. QM stands for quantum mechanics. By the Planck law of QM, NWs having high S/V ratios place NW atoms under high EM confinement thereby denying the atom the heat capacity necessary to increase in temperature. S/V stands for surface-to-volume.
But if so, how do NWs conserve Joule heat?
Since Joule heat cannot be conserved by increasing NW temperatures, QED conserves the surface heat in NWs by creating EM radiation having half-wavelength λ/2 = d standing across the diameter d of the NW. By simple QED, EM radiation is created from surface heat having Planck energy E = hν, i.e., h is Planck’s constant and ν is frequency, ν = (c/n) / λ = c/2nd, where the velocity of light c is corrected for the slower speed in the solid state by the refractive index n of the NW. Once the NW surface heat is expended in forming the standing waves, the EM confinement vanishes and the QED radiation escapes into the surroundings. Since the diameter d is nanoscale, the Planck energy E is created in the EUV allowing the SPR modes in the VIS to be excited by fluorescence. See diverse simple QED applications of the Planck law at the nanoscale at http://www.nanoqed.org/, 2010 – 2017.
Application
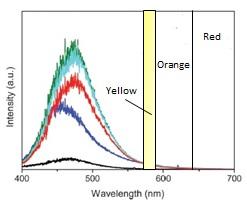
The EUV fluorescence for PET irradiated with laser UV radiation at 325 nm having Planck energy E = 3.82 eV is illustrated in the thumbnail. Although not identical to silver NWs, yellow but not orange and red light is observed in the tail of EUV fluorescence in contrast to that predicted from reflection of ambient light.
UV radiation of PET in the thumbnail may be extended to NWs in PET, i.e., QED induces Joule heat in silver NWs having refractive index n = 1.5 to produce EM radiation E > 3.82 eV in NW diameters d < 108 nm. Typical silver NW diameters d vary from 20 to 150 nm. At the lower end, say d = 20 nm, QED induces Planck energy E = 62.1 eV in the NW that by EUV fluorescence excites SPRs from (350 to 390 nm) at (3.55 and 3.18 eV) in the VIS.
Conclusions
In silver NWs,conservation of Joule heat proceeds by QED inducing EM radiation standing across the NW diameter. Since the NW diameters are nanoscale, EUV radiation in the EUV is created that excites SPRs in the VIS by fluorescence.
Moreover, EUV fluorescence excites all molecules attached to the NW surface such as thin hydrazine [1] coatings, the latter corresponding to the C-N stretching mode 2250–2400 /cm occurring in the IR at 4.3 microns. But the hydrazine coating thickness reduces the SPR, and if too thick alters TCE performance.
Classical physics that assumes NWs have heat capacity [3] is not applicable at the nanoscale. Moreover, thermal heat loss from NWs by the Stefan-Boltzmann law should be superseded at the nanoscale by QED induced EM radiation as NW temperatures are precluded by QM.
References
[1] Y. Cheon, et al., “Reduced yellowing of silver nanowire transparent conductive electrodes by simple hydrazine treatment,” AIP Advances 7, 025215, 2017.
[2] E. Rebollar, et al., “Physicochemical modifications accompanying UV laser induced surface structures on poly(ethylene terephthalate) and their effect on adhesion of mesenchymal cells,”Phys. Chem. Chem. Phys., 16, 17551, 2014.
[3] S. Sorel, et al., “Relationship between Material Properties and Transparent Heater Performance for Both Bulk-like and Percolative Nanostructured Networks,” ACS Nano, 8, 4805–4814, 2014.